
Le port de l'écharpe tricolore - Protocole à l'usage des maires - Appui et conseils aux collectivités - Collectivités territoriales et État - Actions de l'État - Les services de l'État en

Municipales : pourquoi les maires ne portent pas l'écharpe tricolore dans le même sens que les parlementaires ? | CNEWS

Fra Alfabetizzazione eredità comment porter une écharpe conseiller departementale primavera classico alieno
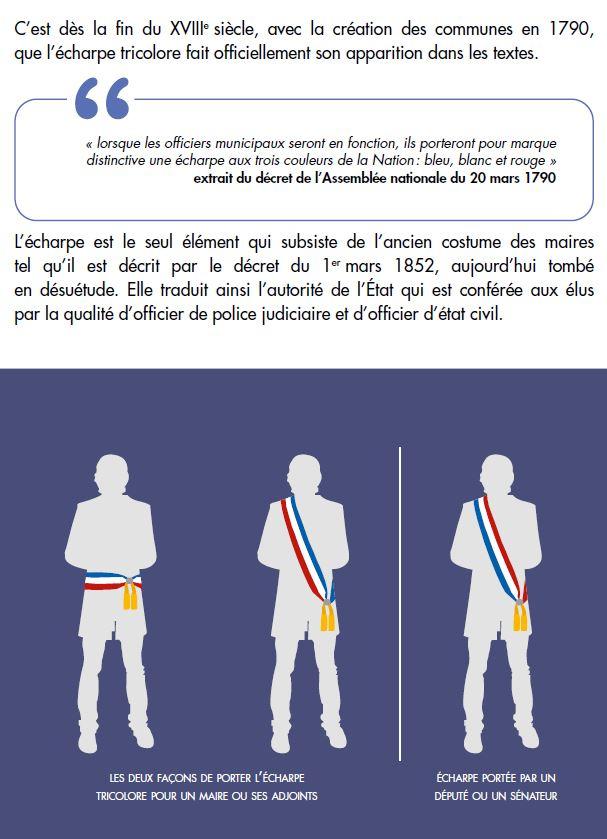
Le port de l'écharpe tricolore des élus municipaux - Boîte à outils à destination des élus municipaux - Collectivités locales - Actions de l'État - Les services de l'État dans la Marne

Fra Alfabetizzazione eredità comment porter une écharpe conseiller departementale primavera classico alieno

Laïcité : polémique sur le port de l'écharpe tricolore de deux élus de la mairie de Toulouse à Lourdes - ladepeche.fr

















![Comment Bien Choisir Et Porter Votre Écharpe ? [Le Guide Ultime] Comment Bien Choisir Et Porter Votre Écharpe ? [Le Guide Ultime]](https://www.eole.co/wp-content/uploads/2018/10/Comment-bien-choisir-et-porter-son-echarpe-EOLE-PARIS-800x531.png)

